With the right partner and people, strong inventory management makes everything else in a veterinary practice run smoother – from daily workflows to patient care. Explore our inventory management resources to help strengthen your system.
Explore our inventory management resources to help strengthen your system.
Check out our featured product, the latest news, and our next webinar. Want to explore more resources or see our full calendar of webinars? Visit our Webinars Library to see all on-demand and upcoming sessions.
featured medications

Every formulation in our equine portfolio is designed with care – meeting strict quality standards to deliver consistent, reliable results.
That means you can focus on what truly matters: better outcomes for your patients. Discover all of the medications both of our brands have available.
Inventory Webinar

Nicole Clausen,
CSSGB, CCFP, Founder, Veterinary Care Logistics, Co-Founder, Inventory Ally
Learn five high-impact ways to simplify inventory management while improving accuracy, efficiency, and profitability. Whether you’re new to inventory or looking to refine your systems, you’ll leave with actionable strategies you can implement immediately to make inventory easier and more sustainable.
new therapy for FCGS
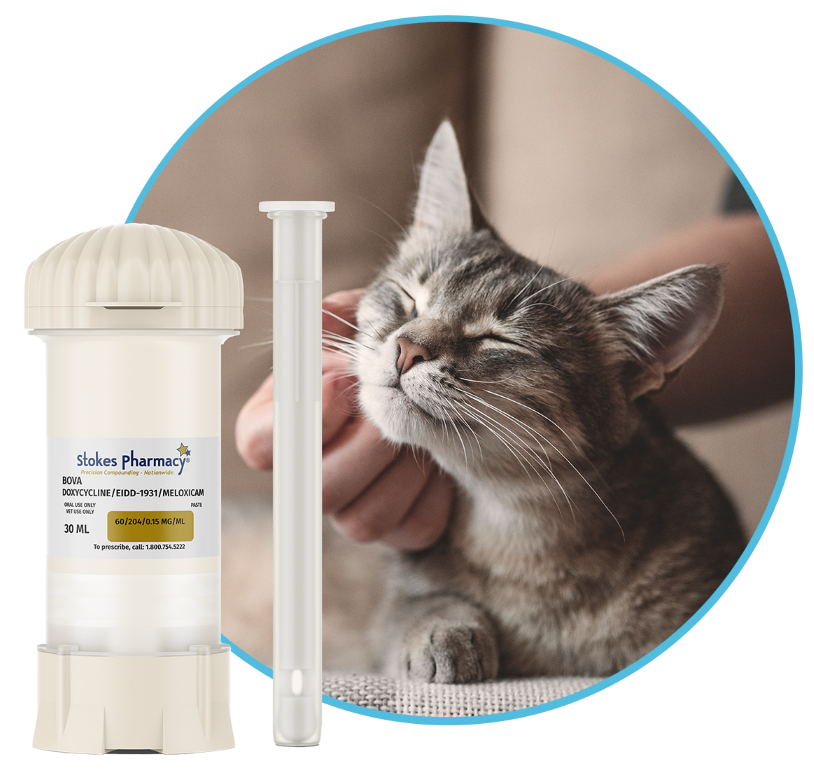
Compounded through our sister division, Stokes Pharmacy
We are proud to share a targeted, feline-specific medication for Feline Chronic Gingivostomatitis that delivers antiviral, anti-inflammatory, and antimicrobial support in one precision-based treatment.
Missed the live webinar? Watch on-demand now!
We manufacture drugs with reliable quality, potency, and sterility in every batch,
so you can fully trust in the medications you give your patients.
One of the largest portfolios of 503B veterinary products
100% manufactured to FDA standards
Medications are verified through stability and sterility testing

Approved for distribution in all 50 states
All drugs ready to ship same day with 1-2 days in transit
From the founding of Stokes Pharmacy to the doors of Epicur Pharma opening, we’re honored that veterinary professionals have continuously trusted our brands. We’re excited to keep serving your practices and patients!
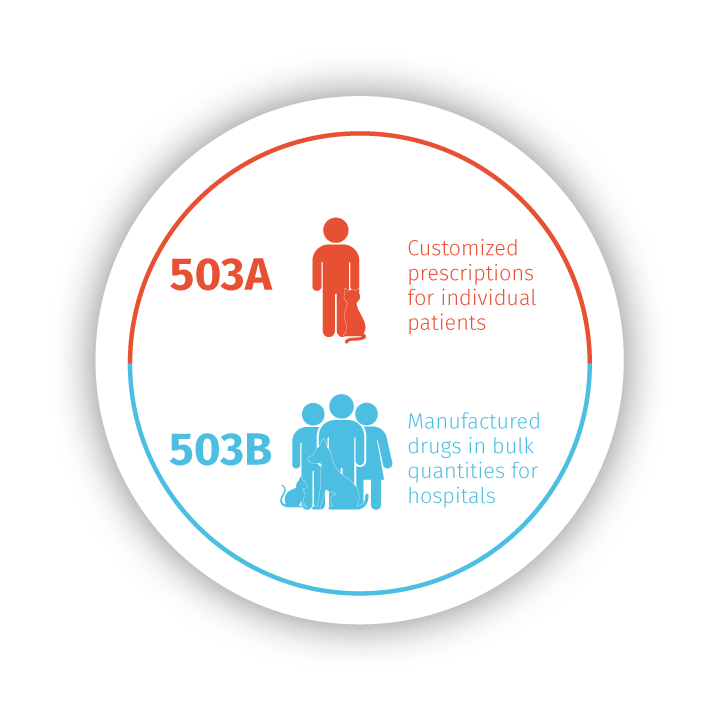
Stokes Healthcare has prepared medications for your individual patients since 1975 through Stokes Pharmacy. Recognizing the evolving needs of veterinary practices as regulations and laws changed, including GFI 256 and USP guidelines, we expanded to include Epicur Pharma to serve the needs of many of your patients with standardized and tested bulk medications.
Each of our brands operates with key differences that impact your patient care and influence treatment plans, but they are all unified in their commitment to quality and improving patient outcomes with medication you trust. You can be confident in your patient care when you partner with us, knowing there is consistency and reliability in every medication.
Learn more about who we are and our commitment to advancing animal care.
Improve compliance and patient outcomes while increasing revenue when you order from Epicur. Whether you’re experiencing shortages or need compounded sterile medications for office-use, Epicur can keep you stocked for critical moments!
New partner? Start your account online or call us at 888-508-5032
If you’re an existing customer, log in to your iFill account to start or manage office orders.
To refill a specific prescription, you can give us a call to place the refill or order it online.
Haven’t used our iFill portal yet? Sign up in a few simple steps to get started!
Get all the latest trends, tips, insights, interviews, and more on our blog!